 For more information on how these figures were generated, see CoolProp.Plots. If we don’t arrive back at the original T,P values, there is a problem in the flash routine in CoolProp. In this figure, we start off with a state point given by T,P and then we calculate each of the other possible output pairs in turn, and then try to re-calculate T,P from the new input pair. The red curve is the maximum temperature curve, and the blue curve is the melting line if one is available for the fluid. A red + is a failure of the flash routine, a black dot is a success. The following figure shows all the flash routines that are available for this fluid. You can also download this figure as a PDF. You can download the script that generated the following figure here: (link to script), right-click the link and then save as… or the equivalent in your browser. They are all results obtained in the form \(Y(T,\rho)\), where \(Y\) is the parameter of interest and which for all EOS is a direct evaluation of the EOS Saturated Vapor Pressure, Boiling Point(dew point), Latent Heat of Vaporization are saturated properties, just enter One parameter to calculate them Saturated. This figure compares the results generated from CoolProp and those generated from REFPROP. R744, co2, CO2, carbondioxide, CARBONDIOXIDE Fluid Information ¶ Recommended Correlations for the Surface Tension of Common Fluids. Surface Tension ¶Ī. Mulero, I. Cachadiña, and M. I. A New Equation of State for Carbon Dioxide Covering the Fluid Region from the Triple Point Temperature to 1100 K at Pressures up to 800 MPa. 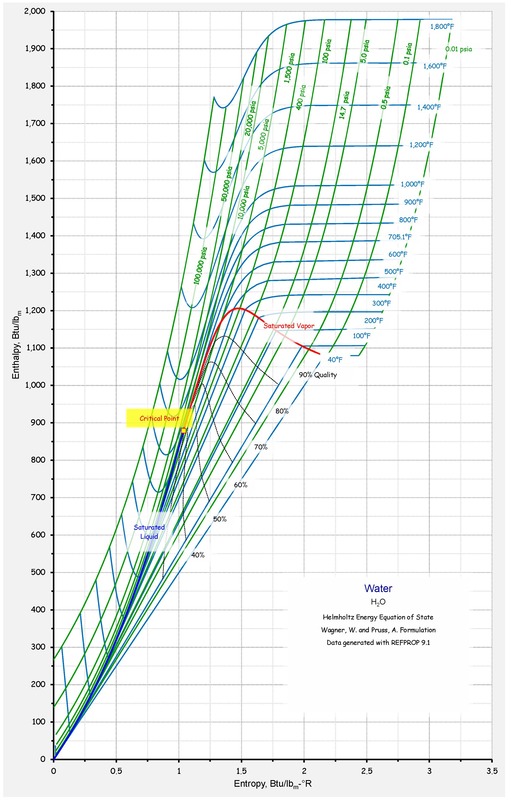
Journal of Physical and Chemical Reference Data, 2017. 
Reference correlation for the viscosity of carbon dioxide. The number of atoms of each element on both sides of CO2 + Ca (OH)2 CaCO3 + H2O is equal which means that the equation is already balanced and no additional work is needed. 
Journal of Physical and Chemical Reference Data, 2016. Step 1: Count the number of each element on the left and right hand sides. Reference correlation of the thermal conductivity of carbon dioxide from the triple point to 1100 k and up to 200 mpa. CarbonDioxide ¶ References ¶ Equation of State ¶ 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
